 
This formula provides meaning to different energy transitions. The mentioned formula leads to the derivation of Rydberg’s formula. V is the frequency of electromagnetic radiation ∆E is the energy that is emitted or absorbed The difference between the two energy levels (assuming E 1, E 2 ) and Planck's constant corresponds to the energy absorbed or emitted: These refer to the excited state of electrons that transition down to a minimum of n=2 and are described by the Balmer equation.Įquations Included In The Bohr Model of Hydrogen Atom 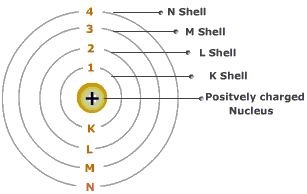
There are four different wavelengths present in a visible spectrum of light in a hydrogen atom. This means that during the transition of electrons to different energy levels, the transmission of light appears. Now, when electrons move between the different energy levels in a hydrogen atom, a spectrum is produced. The maximum energy level an orbital shell can have is the ionization energy of 13.598 eV. 'eV' represents the unit amount of each energy level. Each orbital shell indicates a quantum number 'n' associated with corresponding energy levels. The Neil Bohr atomic model is utilized to explain the working and structural formation of a hydrogen atom and its energy levels. The lowest energy level of an electron is n=1 which is closest to the nucleus and is generally said to be the ground state.īy gaining energy (or photons), an electron can jump to an orbital shell with higher energy, and by losing energy, it can jump to a lower energy level or orbital shell. These numbers are assigned to the shells such as K, L, M, N for 1, 2, 3, 4 respectively. (integers) and are known as quantum numbers. The energy levels of electrons at different orbits are represented by n= 1, 2, 3…. These electrons have definite circular paths known as orbits or shells.Įach of the circular paths has fixed levels of energy and are called orbital shells in terms of this atomic model. Negatively charged electrons revolve around a positively charged nucleus in an atom. The following are the chief characteristics of the atomic model given by Niels Bohr. It was hence proposed that an electron can jump in-between these orbits by gaining or releasing photons (energy). Now, an atom that is in the state of the smallest orbit will be completely stable as the electron will not have a lower orbit it can jump to. The reason for atoms being unstable is that electrons jump to lower orbits causing radiation. Therefore, smaller orbits will have lower energy electrons. To describe Bohr's atomic model, it can be said that the energy levels of the electrons concentrate on the size of the orbits. Niels Bohr atomic theory featured fixed size and energy of the electrons moving in orbits around the nucleus which is charged positively similar to the motion of planets around the sun. Bohr's model was backed up by the classical law of physics and the quantum theory of radiation. This model provided a remedy for the stability problem under the previous Rutherford model of the atom, which displayed a motion where electrons would lose their energy and subsequently spiral into the nucleus due to the emission of electromagnetic radiation by the charged particles. However, the idea of quantized angular momentum has been retained.A quantized shell model of an atom was given by Bohr in 1913 to provide an explanation of how electrons can stably orbit around the nucleus. Modern atomic theory does not allow subatomic particles to be treated in the same way as large objects, and Bohr's reasoning is somewhat discredited. The wavelength of a particle is given by h/ mv, so nh/ mv = 2π r, which leads to mvr = nh/2π. nλ = 2π r, where λ is the wavelength and n a whole number. Each orbit has to have a whole number of wavelengths around it i.e. The idea of quantized values of angular momentum was later explained by the wave nature of the electron. This theory gave good results in predicting the lines observed in the spectrum of hydrogen and simple ions such as He +, Li 2+, etc. 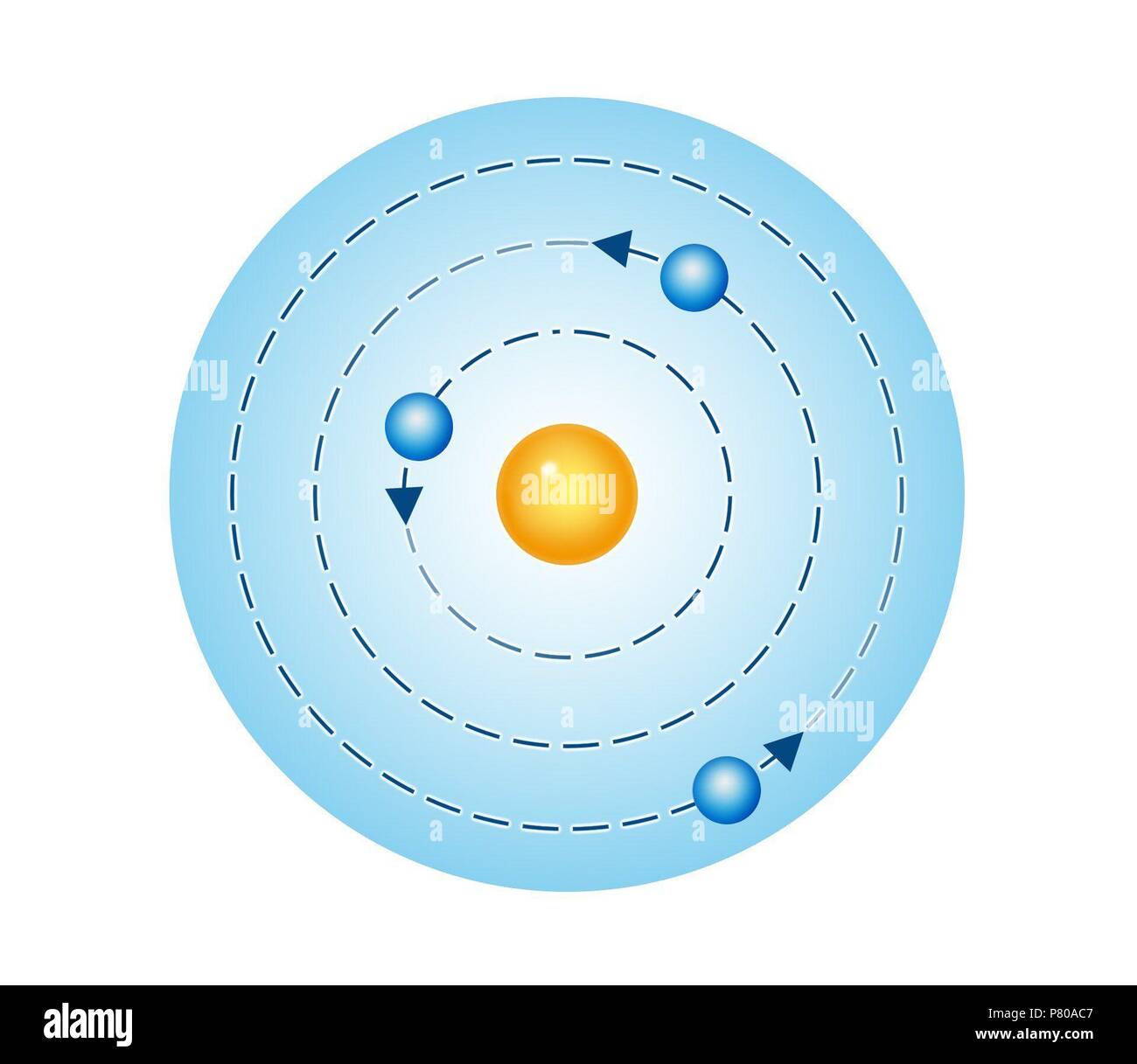
Each permitted value of n is associated with an orbit of different radius and Bohr assumed that when the atom emitted or absorbed radiation of frequency ν, the electron jumped from one orbit to another the energy emitted or absorbed by each jump is equal to hν. can only have certain values, each of which is a multiple of n. This means that the angular momentum is quantized, i.e. Bohr proposed that electrons could only occupy orbits in which this angular momentum had certain fixed values, h/2π, 2 h/2π, 3 h/2π,… nh/2π, where h is the Planck constant. The angular momentum of the electron would then be mvr. He assumed that a single electron of mass m travelled in a circular orbit of radius r, at a velocity v, around a positively charged nucleus. The theory published in 1913 by Niels Bohr to explain the line spectrum of hydrogen. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
